Press Release
27 May 2024
Cessatech reports first patient dosed in final Safety Study 0202 of lead product candidate CT001
- First patient has been dosed in Safety Study 0202 of lead candidate CT001.
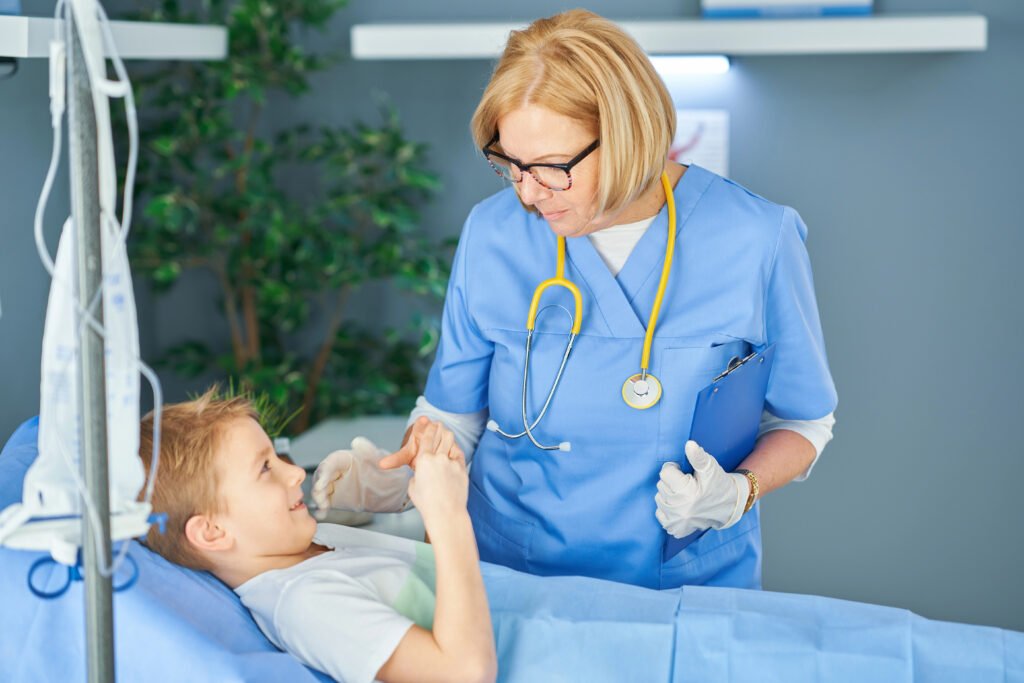
- The Safety Study 0202 is an open-label, prospective study to assess safety, tolerability, analgesic effect, and feasibility of CT001 in 150 pediatric patients with moderate to severe pain, in the emergency setting.
- Cessatech expects the study to be completed by the end of 2024.
27 May – Cessatech A/S announces that the Safety Study 0202 has now been initiated with dosing of the first patient. The trial will assess safety, tolerability, analgesic effect, and feasibility of CT001 in 150 pediatric patients with moderate to severe pain, in the emergency setting.
The pediatric investigation plan (PIP) for CT001 nasal spray has been agreed with the European Medicines Agency, and Safety Study 0202 is part of the clinical development plan for treatment of acute pain in children.
Cessatech announces superior simulated pain efficacy in children
Cessatech announces the final simulated efficacy data in children with CT001 from the abstract that will be presented at the Population Approach Group Europe (PAGE) conference in Rome Italy, during 26–28 June 2024.
- The simulated pain reduction in NRS in children using CT001 was -87%, compared to -52%, -32% and +10% for sufentanil, ketamine and placebo respectively.
- Estimates for the opioid sparing effect of ketamine showed a need for more than double the sufentanil exposure to get the same overall effect seen with the treatment of CT001.
The data supports the earlier reported data from the Dental Study 0205 showing the effectiveness of treatment with CT001. The full simulated data set will be presented at the PAGE conference in Rome 26-28 June.


Jes Trygved
CEO || Cessatech
“We are very pleased with the initiation of this important and final trial for CT001, and the company is now moving into a different stage with last study – great work and effort by all the people involved in the preparation and initiation of this trial, including the countries and sites involved. CT001 is getting closer to the market and its potential patients and the period ahead will be extremely interesting”.
- +45 9387 2309
- jes.trygved@cessatech.com
- www.cessatech.com


